- Research
- Open Access
- Published:
Bacterial ribosomal RNA detection in cerebrospinal fluid using a viromics approach
Fluids and Barriers of the CNSvolume19, Article number:102(2022)
Abstract
Background
In patients with central nervous system (CNS) infections identification of the causative pathogen is important for treatment. Metagenomic next-generation sequencing techniques are increasingly being applied to identify causes of CNS infections, as they can detect any pathogen nucleic acid sequences present. Viromic techniques that enrich samples for virus particles prior to sequencing may simultaneously enrich ribosomes from bacterial pathogens, which are similar in size to small viruses.
Methods
We studied the performance of a viromic library preparation technique (VIDISCA) combined with low-depth IonTorrent sequencing (median ~ 25,000 reads per sample) for detection of ribosomal RNA from common pathogens, analyzing 89 cerebrospinal fluid samples from patients with culture proven bacterial meningitis.
Results
Sensitivity and specificity toStreptococcus pneumoniae(n = 24) before and after optimizing threshold parameters were 79% and 52%, then 88% and 90%. Corresponding values forNeisseria meningitidis(n = 22) were 73% and 93%, then 67% and 100%,Listeria monocytogenes(n = 24) 21% and 100%, then 27% and 100%, andHaemophilus influenzae(n = 18) 56% and 100%, then 71% and 100%. A higher total sequencing depth, no antibiotic treatment prior to lumbar puncture, increased disease severity, and higher c-reactive protein levels were associated with pathogen detection.
Conclusion
We provide proof of principle that a viromic approach can be used to correctly identify bacterial ribosomal RNA in patients with bacterial meningitis. Further work should focus on increasing assay sensitivity, especially for problematic species (e.g.L. monocytogenes), as well as profiling additional pathogens. The technique is most suited to research settings and examination of idiopathic cases, rather than an acute clinical setting.
Key points
We demonstrate that an unmodified viromic next-generation sequencing methodology effectively detects bacterial pathogen ribosomal RNA in cerebrospinal fluid samples. Sensitivity and specificity displayed pathogen dependent variation. Our finding in principle offers avenues for simultaneous detection of neuropathogenic viruses and bacteria.
Introduction
In patients with central nervous system (CNS) infections, rapid identification of the causative pathogen is essential to inform treatment and improve prognosis [1,2]。The differential diagnoses in these patients may include auto-immune disease, non-infectious neurological disease, or non-neurological infection [3,4]。Clinical characteristics fail to adequately differentiate between potential causes, therefore microbiological testing on cerebrospinal fluid (CSF) is the cornerstone of diagnosing CNS infections [5]。目前诊断包括抗原/antibody detection assays, direct microscopy, culture techniques, and quantitative polymerase chain reaction (qPCR). Despite the availability of these tests, in a substantial proportion of patients with a high suspicion of CNS infection, no infectious organism can be identified. Because conventional assays often target specific, common pathogens, uncommon or unknown pathogens may be missed [6,7]。Metagenomic next-generation sequencing (mNGS) is an emerging technique to diagnose CNS infection without targeting specific pathogens [8,9], and is theoretically capable of identifying any pathogen RNA or DNA in samples. As sensitivity and specificity of mNGS assays have yet to match conventional testing, further development is warranted [10,11]。
For detection of viral pathogens, specialized ‘viromic’ mNGS methods have been developed in recent years. Viromic techniques apply mNGS to clinical samples enriched for virus-like particles, minimizing sequencing of host and background nucleic acids in order to maximize sensitivity to viruses. Virus discovery cDNA-amplified fragment length polymorphism (VIDISCA) is one such viromic assay that enables broad detection of known viruses, and has also been applied in the discovery of many novel eukaryotic viruses [12,13,14,15,16]。Viromic assays begin with centrifugation to remove cellular material while retaining virions in supernatant. VIDISCA then treats supernatant with DNase enzymes to remove residual genomic DNA (gDNA), which is unprotected—unlike most viral DNA. Neither step will remove residual mRNA or ribosomes, the latter of which are equivalent in size to small viruses and often highly abundant, depending on the sample type. Consequently, a high proportion of sequence data from clinical specimens can consist of ribosomal RNA (rRNA), which hinders virus detection via competition. To avoid this, VIDISCA incorporates a reverse transcription step using custom hexamer primers that mostly cannot anneal to mammalian rRNA [17], reducing human rRNA sequence reads by over 90% [18]。We previously observed these hexamers still bind to rRNA of some eukaryotic parasites [15], increasing the diagnostic capacity of VIDISCA. So far, the detection of prokaryotic pathogens has not been described. Here, we evaluated the performance of VIDISCA in detection of bacterial rRNA in CSF samples from patients with culture proven bacterial meningitis.
Methods
Sample description
Patients participated in the MeninGene study, a nationwide prospective cohort study of community-acquired bacterial meningitis in the Netherlands, methods of which have been described elsewhere [19,20]。Briefly, patients with a positive CSF culture were identified by the Netherlands Reference Laboratory for Bacterial Meningitis (NRLBM), which receives the cultured pathogen from 85% of bacterial meningitis patients in the Netherlands. The NRLBM notified the researchers, who contacted the treating physician, who subsequently informed patients or their legal representative about the study. Patients could also be included by their treating physician without notification by the NRLBM. All patients or representatives gave written informed consent, and the study was approved by the Medical Ethics Committee of the Amsterdam UMC (METC2013_043). Clinical data were collected using an online case record form and patient outcome was recorded using the Glasgow Outcome Scale [21]。Leftover CSF was stored at treatment centers at − 80 °C and transferred to the Amsterdam UMC biobank facility. For this study, 89 CSF samples with sufficient residual material were selected. Researchers performing library preparation, sequencing and metagenomic analysis were blinded to patient clinical information and the diagnosed pathogen. Subsequently, data were unblinded for optimization of threshold parameters. For controls, previously generated [22] sequencing data from 74 patient CSF samples tested negative in culture for bacteria were included in analysis, approved by a separate decision of the Medical Ethics Committee of the Amsterdam UMC (METC2014_290). These samples were from patients undergoing lumbar puncture for suspected CNS infection, and either viral CNS infection was diagnosed or CNS infection was eventually ruled out.
Library preparation and sequencing
VIDISCA library preparation was performed on CSF as previously described [14]。Briefly, 110 µl of CSF was centrifuged for 10 min at 5000 g, and supernatant was treated with TURBO DNase (Thermo Fisher Scientific) for 30 min at 37 °C. Nucleic acids were extracted using the Boom method [23], followed by reverse transcription primed with non-ribosomal hexamers [17] and second strand synthesis using Klenow fragment (3′ → 5′ Exo-, NEB). After clean-up by phenol/chloroform extraction and ethanol precipitation, dsDNA was digested with Mse1 (NEB) and ligated to sample specific adapters. Size selection with AMPure XP beads (Agencourt) was done to remove small DNA fragments. After a 28-cycle PCR, further size selection was done to retain fragments 200–600 bp long. Library concentrations were analysed using the Qubit dsDNA HS Assay Kit (Thermo Fisher Scientific), and pooled at equimolar concentration. Pool concentration and fragment length distribution were analysed using the Qubit and Bioanalyzer (High Sensitivity Kit, Agilent Genomics) instruments respectively. Sequencing was carried out on the Ion S5 System with the Ion 510 Chip Kit (Thermo Fisher Scientific).
Metagenomic analyses
For bacterial rRNA identification, reads were mapped to the SILVA 138.1 SSU and LSU NR99 rRNA databases [24] using BWA MEM v0.7.17-r1188 [25]。Outputs were processed using the PathoID module of PathoScope v2.0.7 [26], and hits to phylum Chordata were removed. Remaining reads were realigned to the GenBank nt database using BLASTn [27], and hits to the five bacterial pathogens included in this study were counted. Blinded diagnostic predictive ability was explored by selecting the pathogen with the highest read count per sample as the predicted species, and those with equal counts as indeterminate. Unblinded detection performance per pathogen was then measured via sensitivity and specificity calculations. This was repeated varying several threshold parameters to understand their impact and optimize detection performance; parameters were minimum pathogen specific read count (≥ 1 read versus ≥ 10 reads), pathogen read identity to reference (≥ 97% versus 100%), and sample sequencing depth (all samples versus samples ≥ 10,000 reads). The level of human background per sample was estimated by mapping reads to a human rRNA database, subsetted from the aforementioned SILVA database. Hits were realigned to the same database using BLASTn, with reads retained and counted if they matched with 100% nucleotide identity for at least 100 bp.
Sources of bacterial reads (rRNA versus non-rRNA) were assessed by mapping them to genome assemblies ofStreptococcus pneumoniae(GCF_002076835.1),Neisseria meningitidis(GCF_008330805.1), Listeria monocytogenes(GCF_000196035.1), Haemophilus influenzae(GCF_004802225.1), andKlebsiella pneumoniae(GCF_000240185.1). This was done first using original assemblies, and then versions with rRNA genes masked by RepeatMasker v4.1.1 [28]。Reads were curated by realignment to the respective original reference using BLASTn (requiring 100% identity for ≥ 100 bp). The rRNA count was calculated by subtracting the masked assembly read count from the original assembly read count. To determine if non-rRNA was predominately from mRNA or intact gDNA, the proportion mapping to reference coding sequences from each pathogen was calculated, with quality filtration as above. The proportion of coding sequence reads was compared with the gene density of each genome (coding sequence length/total genome length), since this represents the expected proportion of coding sequence reads derived from randomly sequenced pure gDNA.
Detection of viruses was done using a simplified version of a previously published workflow [29]。Reads were aligned to a database of viral proteins, with hits realigned to the GenBank nt database using BLASTn. Those aligning to non-viral sequences were removed as false positives, while those aligning either to viruses or to no reference were retained for manual examination. Reads from viruses of interest were aligned to respective reference genomes (Enterovirus D68: AY426531.1, HIV-1: AF286365.1) in Geneious v4.8.5 (https://www.geneious.com) to visualize genomic position and coverage. HIV-1 reads were subtyped using the Los Alamos National Laboratory HIV BLAST tool (www.hiv.lanl.gov).
Statistical testing
We explored whether clinical characteristics were associated with detection of bacteria using Fisher’s exact tests for dichotomous data or Mann–Whitney U tests for continuous data. Sequencing depth was correlated to clinical characteristics using Spearman’s rank correlation. A p-value of < 0.05 was considered statistically significant.
Results
Patient characteristics
Between 2006 and 2022, 2705 patients with bacterial meningitis were included in the MeninGene study. We selected 89 patient samples from this cohort for VIDISCA analysis (Additional file2: Table S1). Culture based pathogen diagnoses wereS. pneumoniae(n = 24),N. meningitidis(n = 22),L. monocytogenes(n = 24),H. influenzae(n = 18), andK. pneumoniae(n = 1). Thirty-nine patients were female (44%) and the median age was 58 years (interquartile range (IQR) 34–67). Nine patients (10%) were being treated with antibiotics at the moment of presentation. The most frequent symptoms on presentation were fever in 72 patients from 81 case record reports (89%) and headache in 65 patients from 81 reports (80%). An altered mental status, defined as a Glasgow Coma Scale (GCS) score < 14, was seen in 47 patients (53%). Ten patients (11%) were in a comatose state (GCS < 8) upon presentation. Median number of CSF leukocytes was 2560/mm3(IQR 768–5680) with a median CSF total protein concentration of 3.1 g/L (IQR 1.8–5.5). Seventy-three patients (82%) had a favorable outcome, defined as a score of five on the Glasgow Outcome Scale at discharge, and six died (7%). The negative controls consisted of 38 patients with viral CNS infection and 36 patients with initial suspicion of CNS infection, eventually ruled out.
VIDISCA performance in bacterial diagnostics
All 89 CSF samples successfully yielded VIDISCA sequencing reads, with a median of 24,706 (IQR 10,151–40,320). From this we took the lower quartile value (rounded to 10,000 reads) as a threshold sequencing depth, to assess whether bacterial detection was affected by total sequencing depth. Under permissive parameters (any read depth, ≥ 1 pathogen specific read, and ≥ 97% read identity) bacterial pathogen reads were identified in 65 of 89 samples (73%), though only 43 contained reads from a single species. Under strict parameters (sample total read depth ≥ 10,000, ≥ 10 pathogen specific reads, and 100% read identity) 37 of 67 samples (55%) met the pathogen read threshold. Selecting the pathogen with the highest read count per sample, for permissive parameters the culture diagnosed pathogen was correctly predicted for 45 of 65 (69%) samples with bacterial reads, or 51% of all 89 samples. In 19 samples an incorrect pathogen was predicted (21% of 89), all of which were predicted asS. pneumoniae, showing this pathogen carries a particular risk of false positive identification. The final sample with bacterial reads was indeterminate. Under strict parameters the culture diagnosed pathogen was correctly predicted in 34 of 37 (92%) samples, or 38% of all 89, with incorrect prediction for 3 of 89 (3%) samples.
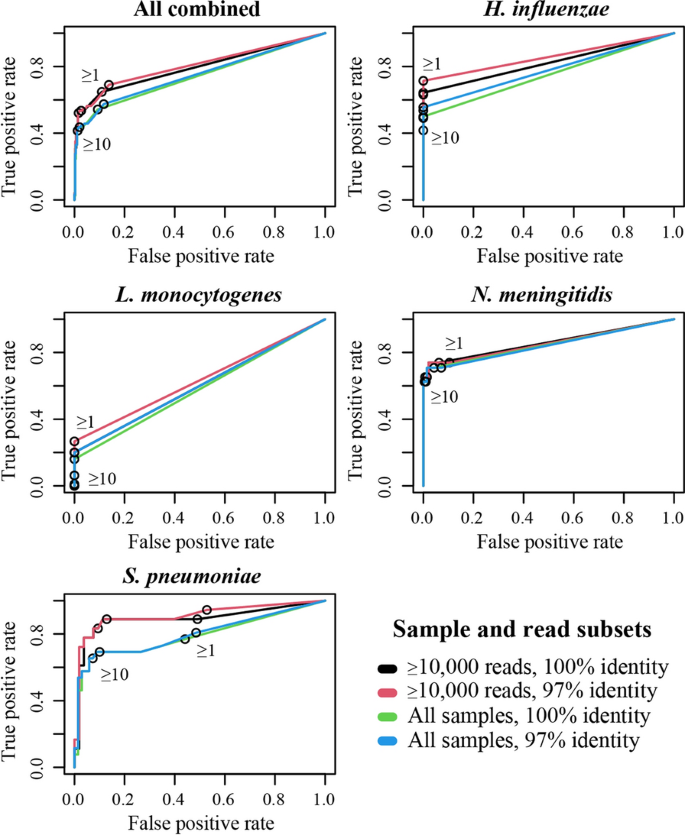
We next explored the impact of threshold parameters on overall and per pathogen diagnostic performance. Alignment identity cut-off, sequencing depth, and pathogen specific read count impacted test diagnostic accuracy, with overall sensitivity to any pathogen ranging from 40% (30–51% CI) to 69% (56–79% CI), and specificity from 87% (82–90% CI) to 99% (97–100% CI; Fig.1, Additional file2: Table S2). For individual pathogens, there was high sensitivity toS. pneumoniae,N. meningitidis, andH. influenzaeacross parameters, and poor sensitivity toL. monocytogenes(Fig.1, Table1). The singleK. pneumoniaepositive sample was successfully detected across all parameter thresholds. Specificity was high across pathogens, with the exception of certain parameter thresholds forS. pneumoniae. Diagnostic performance was higher when only samples with ≥ 10,000 reads were considered, driven by increased sensitivity at low specificity cost, though the impact varied from minimal (e.g.N. meningitidis) to large (e.g.S. pneumoniae) (Fig.1). For minimum pathogen read count, requiring ≥ 10 reads reduced false positive rates forS. pneumoniaefrom ~ 50% to ~ 10% when compared to ≥ 1 read (Fig.1). The impact was lower forL. monocytogenes, H. influenzae,andN. meningitidis,which already had low false positivity rates at the ≥ 1 read threshold (≤ 11%). A ≥ 1 read threshold increased sensitivity for all pathogens compared with ≥ 10 reads, particularly forL. monocytogenesandH. influenzae. Alignment identity requirement had minimal overall impact, though in some cases the ≥ 97% cut-off increased sensitivity compared to the 100% cut-off at low to no specificity cost (Fig.1), leading us to select this as the universal cut-off.
Receiver operating characteristic curves showing VIDISCA diagnostic performance with various parameters set. All 89 samples are analysed together (top left), and then separately by pathogen.K. pneumoniaeis not shown, because n = 1. Subset key refers to total sequencing depth required for sample inclusion, and minimum read alignment identity to a bacterial reference sequence (for ≥ 100 nt). Numbers within charts refer to minimum number of pathogen reads identified for a sample to be called positive
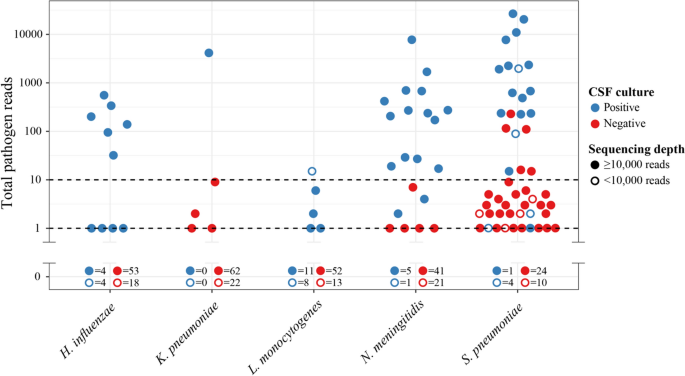
To further understand the differences in diagnostic performance between pathogens, we produced a scatterplot of individual rRNA read counts for each, including all samples (Fig.2). This highlighted a universally low pathogen read count forL. monocytogeneswith a maximum of 15 reads, which likely contributes to the 21% sensitivity we observed (5 of 24 positive samples detected). Notably, 37% ofL. monocytogenesculture positive samples were low-depth (< 10,000 reads), compared to a cohort average of 25%. BothL. monocytogenesandH. influenzaehad zero false positive detections, while bothK. pneumoniaeandN. meningitidishad five, all with < 10 reads (Fig.2).S. pneumoniae经常被发现在样本病人with bacterial meningitis caused by different pathogens, although 26 of 31 (84%) of these had < 10 reads and all were < 250 reads. Analysis of control CSF samples revealed similar false positive patterns (Additional file1: Figure S1), with noL. monocytogenesfalse positives, though this time four falseH. influenzaedetections were made, all with < 10 reads. Six and twoN. meningitisandK. pneumoniaefalse positives were found respectively (all < 10 reads). AgainS. pneumoniaecarried the highest false positive risk, with 28 false positives, 26 of which had < 10 reads, and all of which had < 19.
Associations between clinical data and VIDISCA results
从探索性测试,我们发现检测的e CSF culture diagnosed bacterium by VIDISCA was associated with no antibiotic treatment prior to presentation [49 of 78 (63%) versus 2 of 9 (22%); p = 0.03]. Detection by VIDISCA was lower in patients on immunosuppressive therapy (2 of 12 (17%) versus 48 of 79 (61%); p = 0.004), and higher in patients presenting with an altered mental status [GCS score < 14; 32 of 47 (68%) versus 19 of 42 (45%)] or a comatose state [GCS score < 8; 9 of 10 (90%) versus 42 of 79 (53%); p = 0.04]. GCS score was lower in cases accurately detected by VIDISCA, with 12 (IQR 9–15) versus 15 (IQR 12–15; p = 0.05). The level of C-reactive protein (CRP) was higher in cases detected by VIDISCA, with 206 mg/L (IQR 101–353) versus 91 mg/L (IQR 41–148; p = 0.003). The number of CSF leukocytes did not differ between groups, but CSF protein was higher in VIDISCA detected cases, with 4.1 g/L (IQR 2.9–6.0) versus 2.1 g/L (0.9–3.7; p < 0.001). No correlations between raw read count and clinical variables were found. Raw read count also did not correlate with human rRNA read count as a percentage of the total (Spearman’s rho = 0.16, p = 0.14), but the latter was weakly correlated with CSF leukocyte count (Spearman’s rho = 0.29; p = 0.006).
Source of bacterial nucleic acids
We hypothesized bacterial reads would primarily derive from rRNA and not residual gDNA, due to our library preparation methods. Generally, rRNA did make up the major fraction of detected pathogen reads (Fig.3), especially forL. monocytogenes,N. meningitidis, andS. pneumoniaesamples (all with median values ≥ 87%).S. pneumoniaewas also notable in that some samples had very high rRNA counts, which were not seen in samples containing other bacteria. Across species however, substantial read fractions from bacterial-non-rRNA were also found. In particular,H. influenzaehad a low median value of 36% rRNA, and the singleK. pneumoniaesample had only 10% rRNA, with a high overall bacterial-read count. To determine the likeliest source of non-rRNA reads (mRNA versus residual gDNA) we determined the proportion of non-rRNA aligning to coding sequences of respective pathogen genomes. In most cases this proportion was consistent with a predominately mRNA source, being higher than the expected value for randomly sequenced gDNA (Additional file1: Figure S2). ForH. influenzaehowever, the median proportion was precisely the expected value for gDNA, suggesting DNase treatment or centrifugation may have underperformed in CSF samples containing this species.
Source of bacterial reads detected by whole genome mapping. CSF culture positive samples with at least one filtered read from the respective pathogen are plotted. See also Additional file1: Figure S2
Detection of viruses in CSF
Viral metagenomic analysis identified six CSF samples positive for at least one human virus (Table2). These belonged to four families (Flaviviridae,Anelloviridae,Picornaviridae, andRetroviridae). Reads from Enterovirus D68 (EV-D68, familyPicornaviridae) were identified in one sample, from three regions of the viral genome (Additional file1: Figure S3A). The patient, who was taking prednisone, presented reporting gastrointestinal symptoms for several days and confusion on the day of presentation, followed by an epileptic seizure, and was diagnosed withL. monocytogenesmeningitis. Separately, two distinct HIV-1 reads were detected in the CSF sample of a patient with meningococcal meningitis, both overlapping regions of theEnvgene (Additional file1: Figure S3B). Alignment to subtyped reference genomes showed the reads belonged to HIV-1 subtype B. HIV-1 infection in this patient had been discovered just prior to their admission due to meningitis, and antiretroviral therapy had not yet been started. The patient’s CD4 count was 160 cells/mm3, with a serum viral load of 150,000 copies/mL.
Discussion
In patients with meningitis, no cause is found in approximately 42% of cases [30diagno],要求改进和广泛的敏感stic methods. We demonstrate that the unmodified VIDISCA viromic method, and low-depth sequencing, can detect bacterial pathogens in CSF of patients with bacterial meningitis. Unoptimized predictive accuracy of the culture diagnosed species was low, between 38 and 51% depending on parameters, with inaccurate prediction in 3–21%. Overall sensitivity to bacterial reads was between 40 and 69% depending on threshold parameters, similar to a previous mNGS study [31], suggesting some utility as a diagnostic aid, though more suited to follow-up of undiagnosed meningitis rather than in an acute clinical setting. The performance varied substantially between pathogens, and species-specific parameter optimization improved sensitivity and specificity outcomes. Setting the threshold at a minimum of 10 pathogen reads eliminated allK. pneumoniaeandN. meningitidisfalse positive read detections, and 26 of 31 forS. pneumoniae, while even oneH. influenzaeorL. monocytogenesread was always specific (though four negative control samples did contain up to twoH. influenzaereads). This suggests universal cut-off criteria for diagnosis of pathogens in mNGS assays are suboptimal. Optimization by characterization of individual pathogen mNGS profiles improves the performance and utility of mNGS; however, while this is possible for common etiological agents, it is less feasible for uncommon pathogens.
A lower GCS score on presentation, no use of antibiotics, higher CSF protein levels, and higher blood CRP levels were associated with correct identification of the pathogen by VIDISCA. Several of these variables have previously been associated with increased disease severity [19,32]。Likewise, in both clinical studies and experimental meningitis models higher bacterial loads have been shown to tightly correlate with disease severity [33]。In our study, a higher concentration of bacterial genomic material clearly influenced the likelihood of detection by mNGS. Because CSF is relatively low in genomic and protein background, we hypothesized that higher levels of background nucleic acids (proxied by CSF leukocyte count) would also increase diagnostic success by providing carrier for nucleic acid extraction. However, while CSF leukocyte count was weakly correlated with raw read count, it did not influence diagnostic success.
Apart from bacteria, we were able to identify viruses in a number of CSF samples. Some common and non-pathogenic ones were pegiviruses and members of theAnelloviridae[34,35]。Viruses known to cause CNS infections were found in two samples (EV-D68 and HIV-1), from patients diagnosed withListeriameningitis and meningococcal meningitis respectively.L. monocytogenesinfection of the CNS is commonly preceded by gastrointestinal infection or colonization [36,37], before the pathogen invades the blood and eventually crosses the blood brain barrier [38]。Although EV-D68 is primarily a respiratory virus, it may also present with gastrointestinal symptoms [39,40]。The patient in this case was taking prednisone, and presented reporting gastrointestinal symptoms for several days. The patient appears to have been co-infected by EV-D68 andL. monocytogenes,尽管gastroenterit病原体引起的is is unclear. The clinical significance of HIV-1 detection in one patient is also uncertain, as the virus can often be detected in untreated HIV-1 infection without clinical signs of CNS infection [41], though it also increases the risk of bacterial meningitis by as much as eightfold compared with uninfected individuals [42]。
This study has limitations. We only studied patients with bacterial meningitis confirmed by positive CSF culture, and thus the performance of VIDISCA to discriminate between different causes of infection (viral, bacterial, etc.) cannot be determined in this population. To address this, further studies in patients with suspected CNS infections should be performed. In testing the relationship between clinical variables and pathogen detection, we adopted an exploratory approach using clinical variables already known to correlate with bacterial loads, and therefore did not apply correction for multiple tests. Using data from clinically validated qPCRs would have been preferable, since this would avoid the risks of using both proxy variables and multiple statistical tests. Further, we only studied detection performance for five common pathogens in meningitis. Inclusion of additional pathogens, especially those not found in currently applied clinical rapid tests could be of particular value. Other limitations of this study may not be specific to VIDISCA, for example, the substantial number of false positive detections ofS. pneumoniahas also been observed in multiplex PCR panels [43], probably reflecting a high carriage rate in the population [44]。
In conclusion, we have shown that VIDISCA is capable of detecting bacterial pathogens in CSF, mainly via rRNA. Selective depletion of human rRNA sequences enhances viral detection by VIDISCA [18], and our result implies the same effect likely applies to non-viral pathogens too. This requires rRNA sequences sufficiently distant in sequence to human rRNA, so that reverse transcription primers can anneal. VIDISCA was not developed primarily to detect bacteria, but rather as a low-depth screening method for viruses. As such, the detection rates imply that substantial improvement could be made with optimization, for example by increasing sample sequencing depth. Since VIDISCA selectively depletes gDNA, it is possible DNA-only libraries could be constructed that would enable detection of more bacterial genomic material, as has been done in other mNGS studies [8,9]。The high overall specificity indicates that bacterial analysis of VIDISCA data is applicable in certain circumstances, for example CSF samples already being processed for viral detection and discovery, or for follow-up of idiopathic meningitis cases. The large number of samples processed (40–70) is suited to research and perhaps outbreak settings. However, current sensitivity rates and the turnaround time of between five days to two weeks make it unsuited to the acute clinical setting, where single samples will often require processing. Here, rapid and high performance multiplex assays for common pathogens are more desirable [45]。
Data availability
Not applicable.
References
Brouwer MC, Thwaites GE, Tunkel AR, Van De Beek D. Dilemmas in the diagnosis of acute community-acquired bacterial meningitis. Lancet. 2012;380:1684–92.
van de Beek D, Cabellos C, Dzupova O, et al. ESCMID guideline: diagnosis and treatment of acute bacterial meningitis. Clin Microbiol Infect. 2016;22:S37–62.
Solomon T, Hart IJ, Beeching NJ. Viral encephalitis: a clinician’s guide. Pract Neurol. 2007;7:288–305.
Khatib U, van de Beek D, Lees JA, Brouwer MC. Adults with suspected central nervous system infection: a prospective study of diagnostic accuracy. J Infect. 2017;74:1–9.
Brouwer MC, Tunkel AR, Van De Beek D. Epidemiology, diagnosis, and antimicrobial treatment of acute bacterial meningitis. Clin Microbiol Rev. 2010;23:467–92.
Edridge AWD, Deijs M, Van Zeggeren IE, et al. Viral metagenomics on cerebrospinal fluid. Genes. 2019;10:332.
Whitley RJ, Gnann JW. Viral encephalitis: familiar infections and emerging pathogens. Lancet. 2002;359:507–13.
Wilson MR, Sample HA, Zorn KC, et al. Clinical metagenomic sequencing for diagnosis of meningitis and encephalitis. N Engl J Med. 2019;380:2327–40.
Piantadosi A, Mukerji SS, Ye S, et al. Enhanced virus detection and metagenomic sequencing in patients with meningitis and encephalitis. MBio. 2021;12:e01143-e1221.
Gu W, Deng X, Lee M, et al. Rapid pathogen detection by metagenomic next-generation sequencing of infected body fluids. Nat Med. 2021;27:115–24.
Olausson J, Brunet S, Vracar D, et al. Optimization of cerebrospinal fluid microbial DNA metagenomic sequencing diagnostics. Sci Rep 2022; 12.
van der Hoek L, Pyrc K, Jebbink MF, et al. Identification of a new human coronavirus. Nat Med. 2004;10:368.
de Groof A, Guelen L, Deijs M, et al. A novel virus causes scale drop disease inLates calcarifer. PLoS Pathog. 2015;11: e1005074.
Edridge AWD, Deijs M, Namazzi R, et al. Novel orthobunyavirus identified in the cerebrospinal fluid of a Ugandan child with severe encephalopathy. Clin Infect Dis. 2018;68:139–42.
Kinsella CM, Bart A, Deijs M, et al.EntamoebaandGiardiaparasites implicated as hosts of CRESS viruses. Nat Commun. 2020;11:1–10.
Edridge AWD, Abd-Elfarag G, Deijs M, Jebbink MF, van Hensbroek MB, van der Hoek L. Divergent rhabdovirus discovered in a patient with new-onset nodding syndrome. Viruses. 2022;14:210.
Endoh D, Mizutani T, Kirisawa R, et al. Species-independent detection of RNA virus by representational difference analysis using non-ribosomal hexanucleotides for reverse transcription. Nucleic Acids Res. 2005;33: e65.
de Vries M, Deijs M, Canuti M, et al. A sensitive assay for virus discovery in respiratory clinical samples. PLoS ONE. 2011;6: e16118.
Bijlsma MW, Brouwer MC, Kasanmoentalib ES, et al. Community-acquired bacterial meningitis in adults in the Netherlands, 2006–14: a prospective cohort study. Lancet Infect Dis. 2016;16:339–47.
Lees JA, Ferwerda B, Kremer PHC, et al. Joint sequencing of human and pathogen genomes reveals the genetics of pneumococcal meningitis. Nat Commun. 2019;10:1–14.
Jennett B,债券m .签订后的评估结果ere brain damage: a practical scale. Lancet. 1975;305:480–4.
van Zeggeren IE, Edridge AWD, van de Beek D, et al. Diagnostic accuracy of VIDISCA-NGS in patients with suspected central nervous system infections. Clin Microbiol Infect. 2021;27:631.e7-631.e12.
Boom R, Sol CJ, Salimans MM, Jansen CL, Wertheim-Van Dillen PM, van der Noordaa J. Rapid and simple method for purification of nucleic acids. J Clin Microbiol. 1990;28:495–503.
Quast C, Pruesse E, Yilmaz P, et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 2013;41:D590–6.
Li H. Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. arXiv:13033997v1 [q-bioGN] 2013;
Hong C, Manimaran S, Shen Y, et al. PathoScope 2.0: a complete computational framework for strain identification in environmental or clinical sequencing samples. Microbiome. 2014; 2.
Camacho C, Coulouris G, Avagyan V, et al. BLAST+: architecture and applications. BMC Bioinformatics. 2009;10:421.
Smit A, Hubley R, Green P. RepeatMasker Open-4.0. 2013–2015.http://www.repeatmasker.org. 2015;
Kinsella CM, Deijs M, van der Hoek L. Enhanced bioinformatic profiling of VIDISCA libraries for virus detection and discovery. Virus Res. 2019;263:21–6.
McGill F, Griffiths MJ, Bonnett LJ, et al. Incidence, aetiology, and sequelae of viral meningitis in UK adults: a multicentre prospective observational cohort study. Lancet Infect Dis. 2018;18:992–1003.
Xing XW, Zhang JT, Ma YB, et al. Metagenomic next-generation sequencing for diagnosis of infectious encephalitis and meningitis: a large, prospective case series of 213 patients. Front Cell Infect Microbiol. 2020;10:88.
van de Beek D, de Gans J, Spanjaard L, Weisfelt M, Reitsma JB, Vermeulen M. Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med. 2004;351:1849–59.
Brandt CT, Lundgren JD, Lund SP, et al. Attenuation of the bacterial load in blood by pretreatment with granulocyte-colony-stimulating factor protects rats from fatal outcome and brain damage duringStreptococcus pneumoniaemeningitis. Infect Immun. 2004;72:4647–53.
Yu Y, Wan Z, Wang JH, Yang X, Zhang C. Review of human pegivirus: prevalence, transmission, pathogenesis, and clinical implication. Virulence. 2022;13:323–40.
Kaczorowska J, Van Der Hoek L. Human anelloviruses: diverse, omnipresent and commensal members of the virome. FEMS Microbiol Rev. 2020;44:305–13.
Luthe SK, Sato R, Maeda T, Takahashi K. Case report:Listeria monocytogenesmeningitis preceded by acute cholangitis. BMJ Case Rep. 2017;
Salamina G, Dalle Donne E, Niccolini A, et al. A foodborne outbreak of gastroenteritis involvingListeria monocytogenes. Epidemiol Infect. 1996;117:429–36.
Disson O, Lecuit m .针对中枢神经us system byListeria monocytogenes. Virulence. 2012;3:213–21.
Wang H, Diaz A, Moyer K, et al. Molecular and clinical comparison of Enterovirus D68 outbreaks among hospitalized children, Ohio, USA, 2014 and 2018. Emerg Infect Dis. 2019;25:2055–63.
Sooksawasdi Na Ayudhya S, Laksono BM, van Riel D. The pathogenesis and virulence of enterovirus-D68 infection. Virulence. 2021;12:2060–72.
Singer EJ, Valdes-Sueiras M, Commins D, Levine A. Neurologic presentations of AIDS. Neurol Clin. 2010;28:253.
van Veen KEB, Brouwer MC, van der Ende A, van de Beek D. Bacterial meningitis in patients with HIV: a population-based prospective study. J Infect. 2016;72:362–8.
Tansarli GS,查宾KC。诊断测试精度of the BioFire® FilmArray® meningitis/encephalitis panel: a systematic review and meta-analysis. Clin Microbiol Infect. 2020;26:281–90.
Satzke C, Turner P, Virolainen-Julkunen A, et al. Standard method for detecting upper respiratory carriage ofStreptococcus pneumoniae: updated recommendations from the World Health Organization Pneumococcal Carriage Working Group. Vaccine. 2013;32:165–79.
Trujillo-Gómez J, Tsokani S, Arango-Ferreira C, et al. Biofire FilmArray Meningitis/Encephalitis panel for the aetiological diagnosis of central nervous system infections: a systematic review and diagnostic test accuracy meta-analysis. eClinicalMedicine. 2022; 44:101275. Available at:http://www.thelancet.com/article/S2589537022000050/fulltext. Accessed 31 October 2022.
Acknowledgements
Computational work was carried out on the Dutch national e-infrastructure with the support of SURF Cooperative.
Funding
This work was supported by a European Union Horizon 2020 research and innovation program Grant [Marie Skłodowska-Curie agreement No. 721367 (HONOURs) to Lia van der Hoek], the Netherlands Organization for Health Research and Development (ZonMw; NWO-Vidi Grant [Grant number 917.17.308 to Matthijs C. Brouwer], NWO-Vici-Grant [Grant number 918.19.627 to Diederik van de Beek]) and the European Research Council (ERC Consolidator grant [Grant 101001237 to Matthijs C. Brouwer]).
Author information
Authors and Affiliations
Contributions
Conceptualization, LvdH, MCB, CMK, AWDE; Methodology, AWDE, CMK, IEvZ, LvdH, MCB; Software, CMK; Clinical data and sample collection, MCB, DvdB, IEvZ; Formal analysis, AWDE, CMK, IEvZ; Investigation, AWDE, CMK, IEvZ, MD; Resources, MCB, LvdH, DvdB; Writing—Original Draft, CMK, LvdH, MCB, AWDE, IEvZ; Writing—Review & Editing, all authors; Visualization, AWDE, CMK; Supervision, LvdH, MCB; Funding acquisition, LvdH, MCB, DvdB. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Medical Ethical Committee of the Amsterdam UMC (METC2013_043 and METC2014_290). All patients or their legal representatives gave written informed consent.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
检测的假阳性读取控制脑脊液amples. For each pathogen, read detection across all 74 samples is plotted. Dotted lines denote the pathogen read count thresholds utilised in this study.Figure S2.Source of bacterial non-rRNA reads detected by whole genome mapping. The proportion of non-rRNA reads derived from bacterial coding sequence (CDS) is plotted per sample. CSF culture positive samples with at least one filtered non-rRNA read from the respective pathogen are plotted. Dotted lines represent the expected proportion of CDS reads if pure gDNA were randomly sequenced (gene density in the genome expressed as a proportion). In contrast, the expected proportion of CDS reads if pure mRNA were randomly sequenced is 1.00. Low outlier samples all had <10 total non-rRNA reads.Figure S3.CSF detection of neuropathogenic viruses. A) Enterovirus D68 (EV-D68) in a patient with CSF culture confirmedListeriameningitis. Reads were aligned to EV-D68 strain Fermon (AY426531.1), with the position of the polyprotein open reading frame shown in black. Read matches to references are light grey, while mismatches are black. B) HIV-1 subtype B in a patient with CSF culture confirmedN. meningitidisinfection. Reads were aligned to HIV-1 isolate WR27 (AF286365.1), with the positions of structural genes shown in black. Read matches to references are light grey, while mismatches are black.
Additional file 2: Table S1.
Baseline characteristics of included patients. Values are n/N (%) or median (interquartile range).Table S2.VIDISCA performance for bacterial diagnostics.
Rights and permissions
Open AccessThis article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visithttp://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Kinsella, C.M., Edridge, A.W.D., van Zeggeren, I.E.et al.Bacterial ribosomal RNA detection in cerebrospinal fluid using a viromics approach.Fluids Barriers CNS19, 102 (2022). https://doi.org/10.1186/s12987-022-00400-5
Received:
Accepted:
Published:
DOI:https://doi.org/10.1186/s12987-022-00400-5
Keywords
- Bacterial meningitis
- Pathogen detection
- beplay靠谱
- Viromics
- Metagenomics